|
On breaking down the compound, the smallest entity so obtained is called a molecule of the compound. Therefore, a compound does not exhibit the properties of its constituent elements. Compound formation always takes place as a result of a chemical reaction. Metalloids are the elements that have properties that are in between those of metals and nonmetals.Įxamples include boron, silicon, arsenic, antimony, germanium, etc.Ī compound is a pure substance that is formed by the chemical combination of two or more elements in a fixed proportion by mass. The physical properties of non-metals include brittleness, comparatively lower tensile strength, non-luster, non-conductivity or insulation, lower melting and boiling points, etc.Įxamples include hydrogen, helium, chlorine, iodine, sulfur, phosphorus oxygen, etc. Non-metals are the elements that have the tendency of gaining electrons in order to gain stability, that is, they exhibit electronegativity. The physical properties of metals include hardness, high tensile strength, luster, conductivity, high melting and boiling points, etc.Įxamples include sodium, potassium, iron, calcium, magnesium, lead, tin, etc. Metals are the elements that have the tendency of losing electrons in order to gain stability, that is, they exhibit electropositivity. Some commonly used symbols of elements are listed below.ĭepending on the properties, elements can be classified into metals, nonmetals, and metalloids. To represent an atom of an element, we use symbols. They always tend to combine with other elements in order to attain stability. 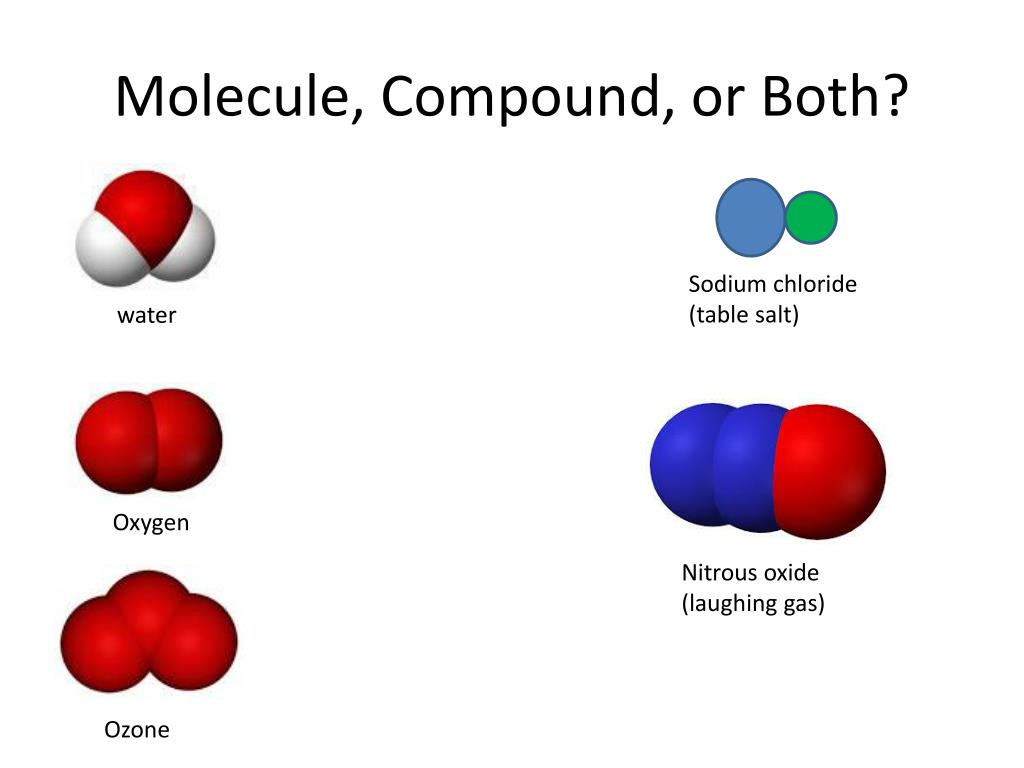
Atoms are highly unstable, therefore do not exist independently. The smallest entity of an element is called an atom. Every element has a fixed place in the periodic table, depending on its properties. To date, more than 119 elements have been discovered and many more are in the process of being discovered. All matter is fundamentally composed of elements. 
In chemistry, an element is a substance that cannot be further broken down into a simpler substance by using common chemical methods. Depending on their composition, they can further be classified into homogeneous and heterogeneous. Impure substances are also called mixtures. Pure substances can further be classified into elements and compounds. Impure substances are the ones that can have varying compositions, and as such do not exhibit a fixed set of properties. Pure substances are the ones that have a fixed chemical composition. The matter around us can be categorized into pure matter and impure matter. It is therefore imperative to classify and categorize them so that their properties can be easily studied, analyzed, and understood. All around us, we see various kinds of substances and materials. 
Chemistry is the study of various kinds of matter and its properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
